Motor neurons are important cells in our body that send signals to our skeletal muscle that allow us to do activities like walk, speak, swallow, and even breathe. Motor neuron diseases like amyotrophic lateral sclerosis (ALS) destroy motor neurons. When muscles don’t receive normal signals from our motor neurons, the muscles atrophy (weaken and shrink) and we lose the ability to do those activities over time.
Currently, there are no treatments available to fully prevent the progression of motor neuron diseases.
Researchers around the world are studying motor neuron diseases to better understand how to treat them. One challenge currently slowing progress in this field is that there are few reliable in vitro models of motor neuron diseases.
I developed a system for generating large quantities of mature human motor neurons in vitro that can be used to study all types of motor neuron diseases, as well as potential therapies, in the lab. The full protocol was published in 2020 in Stem Cell Reports here.
What do motor neurons actually look like?
The soma, the round-ish cell body of a motor neuron, is typically about 10 micrometers in humans, which is large enough to see through a microscope. For our paper, we commissioned an artist to sketch motor neurons at various stages of development based on microscopy images:
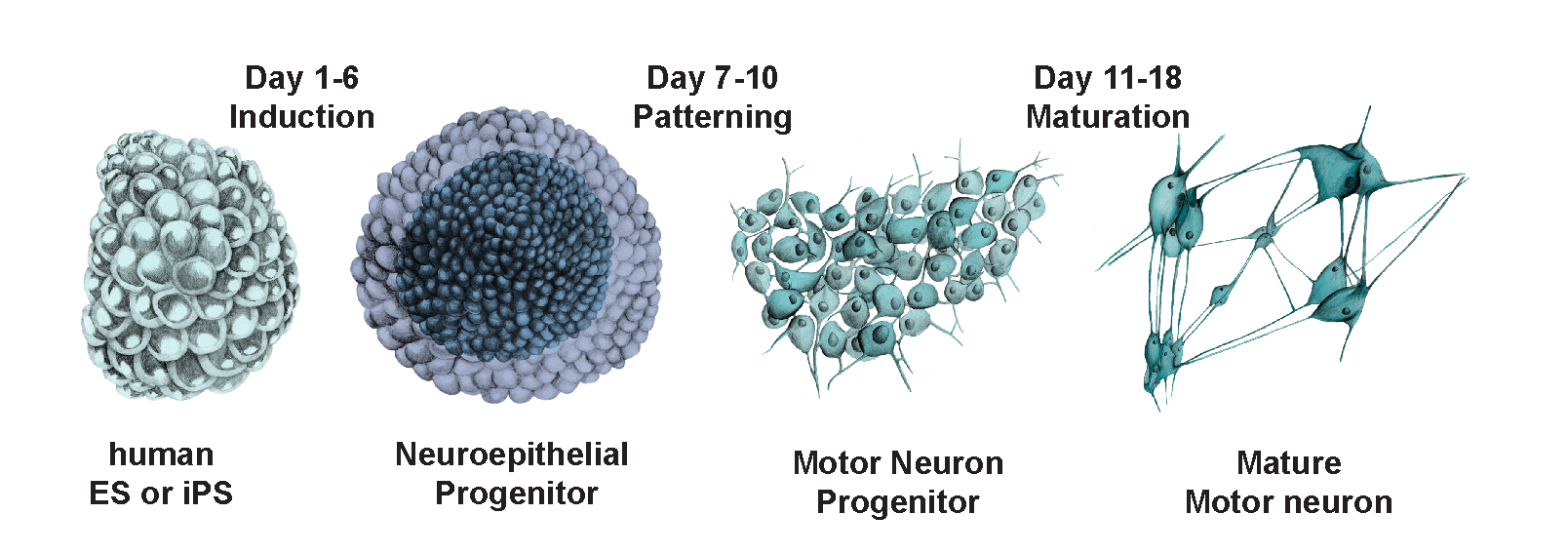
In biology, form follows function. As a motor neuron matures, it develops an axon, which is used for its main function of transmitting signals long distances across the body.
To grow motor neurons in vitro in a way that accurately represents how they grow in vivo, we designed a protocol for guiding them through the developmental stages above.
The starting material for this protocol can be either induced-pluripotent stem cells (iPS) or human stem cells (ES). First, we place cells in media (cell food!) and allow them to grow and replicate, forming clusters of about 5000 cells each.
Next, we provide them with various molecules which serve as cues to progress through the developmental stages. Within about 18 days, the undifferentiated cells we started with will have become mature motor neurons which can form connections and send signals to each other (see image below). Before our protocol, this process previously took at least a month and sometimes much more!
What our cells actually looked like in the dish
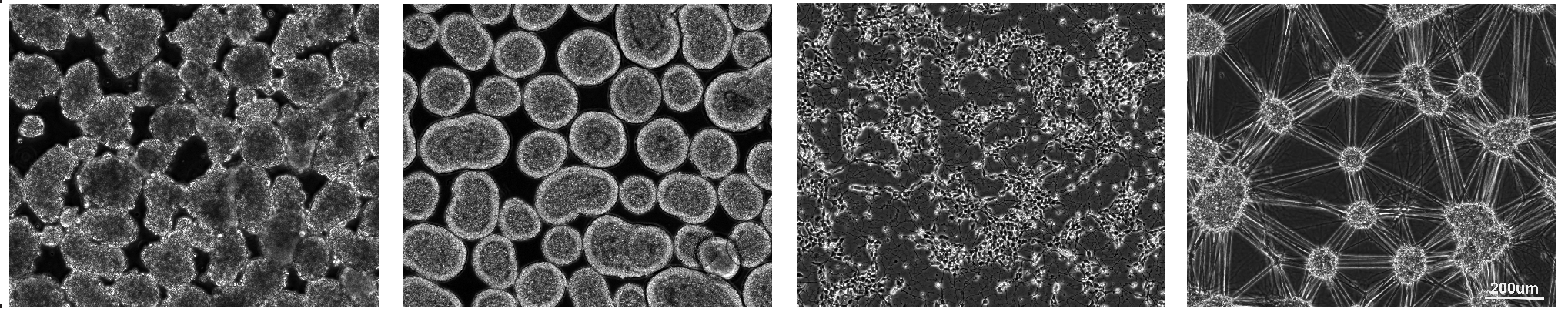
Motor neuron cells developed using our protocol at day 0, 7, 13, and 20 days (left to right).
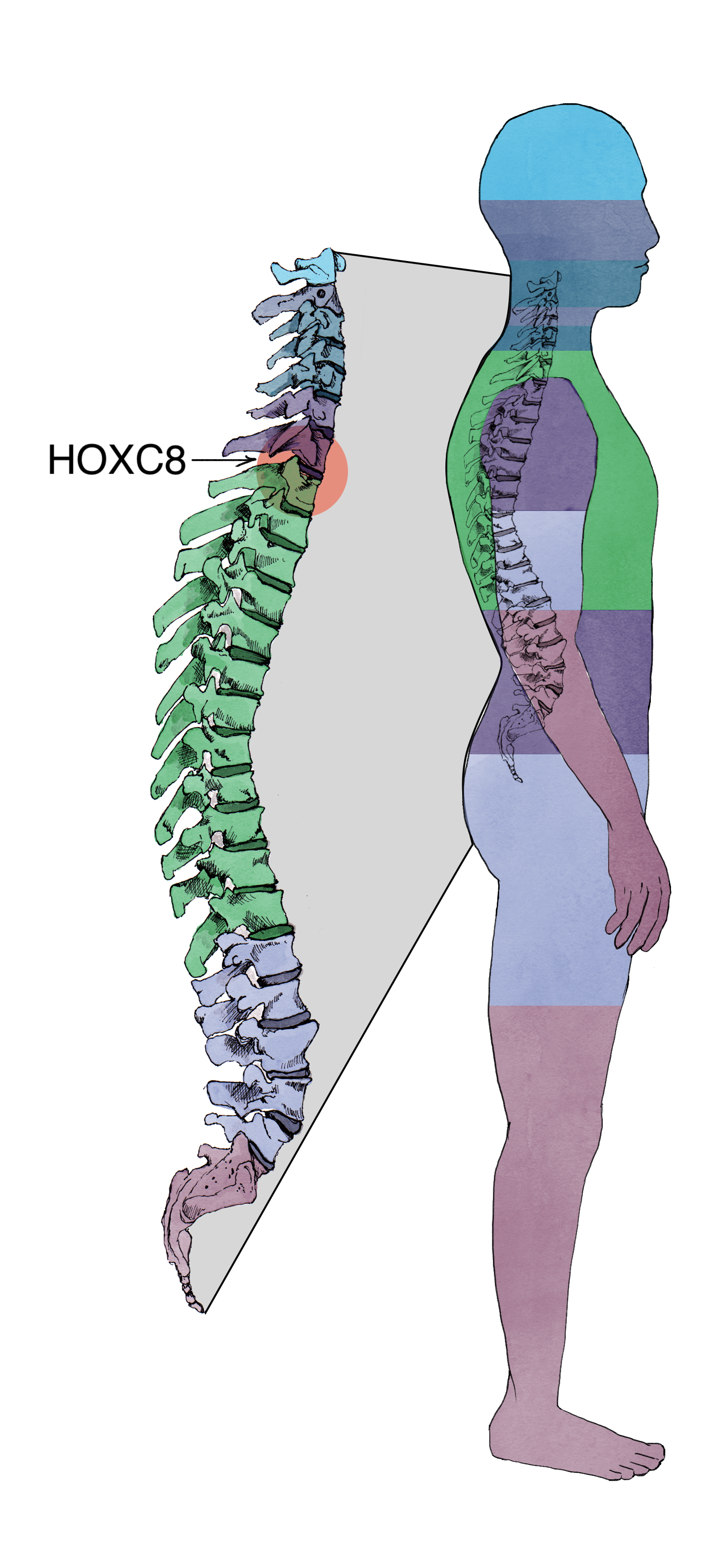
It may sound funny, but when we set out to do this experiment, we weren’t sure which types of motor neurons the protocol would actually produce. To our surprise, the motor neurons we created expressed the same gene (HOXC8) as motor neurons located specifically in the thoracic/lumbar section of the spinal cord. In the human body, these motor neurons innervate the legs, making them a perfect model system to study motor neuron diseases.
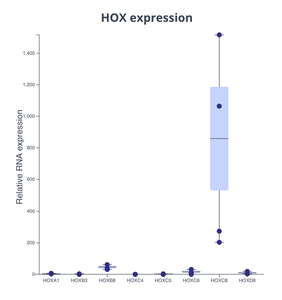
Generated motor neurons are specific to the HOXC8 region of the spinal cord.
Barplot showing HOX gene expression in generated motor neurons. HOXC8 stands out clearly as the only gene expressed by these cells. Plot made with Pluto.
Once we had produced the cells that we wanted to study, we decided to subject them to hypoxic conditions (lower than normal oxygen levels) because it has been reported that motor neurons of diabetic patients experience hypoxic conditions due to poor blood circulation in the periphery (i.e. the patient’s feet). To identify genes and pathways affected by hypoxia exposure, we used RNA-sequencing (RNA-seq).
To take it a step further, we wanted to ascertain which of those genes and pathways changed in the soma (cell body) versus their neurites (long projections). To do this, we performed a fractionation step to separate the two. Knowing which RNAs came from the soma versus the neurites can be extremely important in understanding disease progression because transport of RNAs from soma to neurites can be affected and contribute to disease onset and progression.
Separation of cell body and cell extensions
We sequenced 4 different types of samples (n = 3 per condition):
- Soma - normal oxygen levels
- Soma - hypoxia
- Neurites - normal oxygen levels
- Neurites - hypoxia
Because we didn’t have a cloud-based platform at the time, the collaboration and analysis of this data took many weeks, but we were able to identify interesting genes and pathways that were affected by hypoxic conditions.
Fortunately, you don't have to want to wait this long to see your exciting results. Using Pluto, you can upload your experimental data and begin exploring results immediately.
In the cell body, hypoxia induced expression of glycolytic pathway genes among others. This is important because it suggests that the cells switched from using oxidative phosphorylation (OXPHOS) pathway to produce energy to glycolysis which is much less efficient (2ATP are produced during glycolysis and 36 during OXPHOS). Additionally, mitochondrial translation genes were increased in the soma of hypoxic samples. Mitochondria is a critical cellular organelle responsible for generating most of the energy cells for survival and function, including using OXPHOS for energy production. Based on this observation we hypothesized that hypoxic cells have more mitochondria. To test this, we labeled mitochondria in live cells exposed to hypoxic conditions but interestingly found that they had significantly less mitochondria in their neurites than cells in normal oxygen levels (Pluto graph below). Therefore, these data suggest that hypoxia causes a loss of mitochondria in the neurites of motor neurons, which in turn triggers the soma to upregulate genes that encode the mitochondrial components in an attempt to make more mitochondria in response to the loss. The loss of mitochondria further explains the switch from OXPHOS to glycolysis we described above.
Neurites in hypoxic human motor neurons do have lower numbers of mitochondria
Images and barplot showing mitotracker expression. Plot made with Pluto.
In summary, our ability to use powerful tools such as RNA-seq allowed us to discover that loss of mitochondria from the neurites of motor neurons is an important factor contributing to motor neuron diseases and the loss of function seen in motor neurons of diabetic patients. This discovery not only helps with our understanding of disease, but also with our ability to properly target it for best patient outcomes.
References
Hudish LI, Bubak A, Triolo TM, Niemeyer CS, Lorberbaum DS, Sussel L, Nagel M, Taliaferro JM, Russ HA. Modeling Hypoxia-Induced Neuropathies Using a Fast and Scalable Human Motor Neuron Differentiation System. Stem Cell Reports 2020 Jun 9;14(6):1033-1043. doi: 10.1016/j.stemcr.2020.04.003. PMID: 32386561.
Pluto ensures that you not only have fast and reliable results so that you can continue to push the boundaries of knowledge, but that those results are also easily accessible, shareable and publishable right from your browser. Ready to give it a try? Contact us to schedule a personalized demo for your lab.
