This study conducted by a research team at the University of Turku attempts to assess the ability of the mRNA vaccine BNT162b2/Comirnaty by Pfizer-BioNTech to mount a response against 2 of the new variants causing new waves of infections, the B.1.1.7 (alpha) and B.1.351 (beta).
Both mRNA vaccines from Moderna and Pfizer were designed against the spike protein (S1) of the virus. The alpha and beta variants have 8 to 10 amino acid mutations in the spike protein - this study tests whether those mutations render the vaccine less effective against those variants.
To test the efficacy of the vaccine against these variants, the group collected serum samples from 180 health care workers aged 20-65 who received their 2 doses of the Pfizer vaccine 3 weeks apart. The volunteers were mostly female (83%) . The team also collected serum from 50 at-home treated COVID patients. This was used as a comparison group to help assess whether COVID infection was more or less effective at eliciting antibody levels than the vaccine. It is important to note that severe infection does elicit stronger antibody levels than less severe infections such as the group used in this study which did not need hospitalization.
Volunteers that participate in clinical studies like these are critical to advancing scientific knowledge.
The mRNA vaccines have been shown to elicit strong antibody responses against the first COVID variant identified. COVID infection produces antibodies against both the spike protein (S1) and nucleoprotein (N) but vaccines induce only anti-S1 antibodies. The study analyzed the samples of the volunteers before any vaccine dose was administered (day 0) and found that 11 of the 180 subjects had anti-S1 antibodies (Supp Figure 1A) indicating they had a previous COVID infection. These individuals showed a significant increase in their antibody titers after just the first dose of the vaccine.
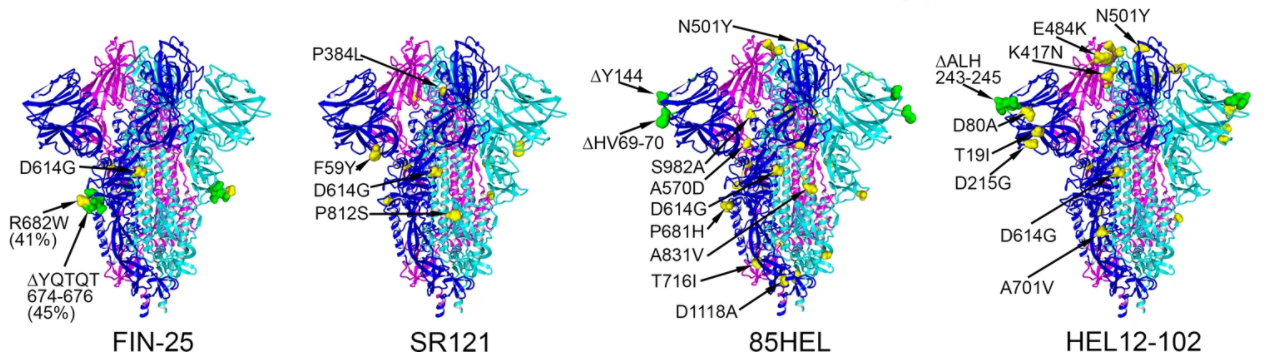
Protein structures of spike protein variants.
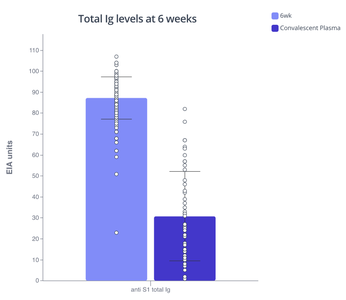
The rest of the volunteers without a prior COVID infection developed varying levels of anti-S1 antibodies after the 1st dose (3 weeks). However, six weeks after the first dose (three weeks after the second dose) all participants elicited high levels of anti-S1 antibodies and their levels were significantly higher than the anti-S1 levels detected in the patient cohort (dark purple).
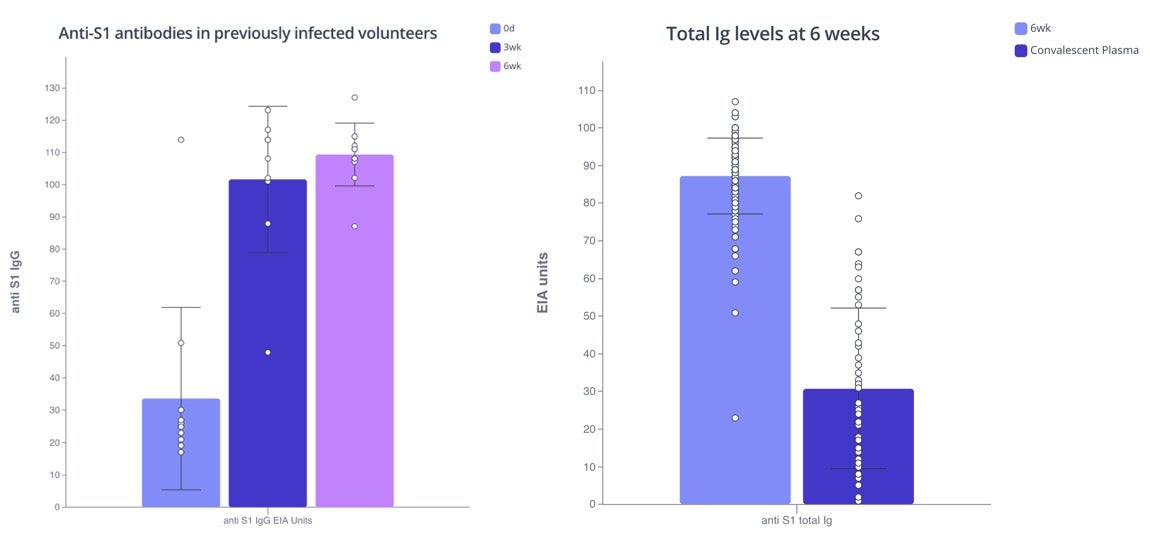
Barplot on left shows increasing levels of antibodies after the 1st dose (3 week) in previously-infected volunteers. Barplot on right shows higher antibody levels after 2nd dose of vaccine (6 week) than after COVID-19 infection without hospitalization. Plots made with Pluto.
Viruses which encode their genetic material in RNA like COVID-19, tend to incur mutations quickly as they are being copied in their hosts. The proteins copying the viral RNA makes mistakes (mutations) which can be either detrimental or beneficial to the virus. COVID-19 mutates at a relatively slow rate compared to other RNA viruses such as HIV.
To assess the efficacy of these antibodies against the new variants, the team used the sera from the volunteers and tested it against four variants circulating in Finland at the time. They found that the response against the alpha variants was strong with 100% of the vaccines having neutralizing antibodies whose levels exceeded those of the convalescent COVID patients. The titers against the beta variant (HEL12-102) were five-fold lower than the others but 92% of vaccines still showed a reasonable antibody response indicating that the vaccine is still able to elicit a good response and protection against the beta variant.
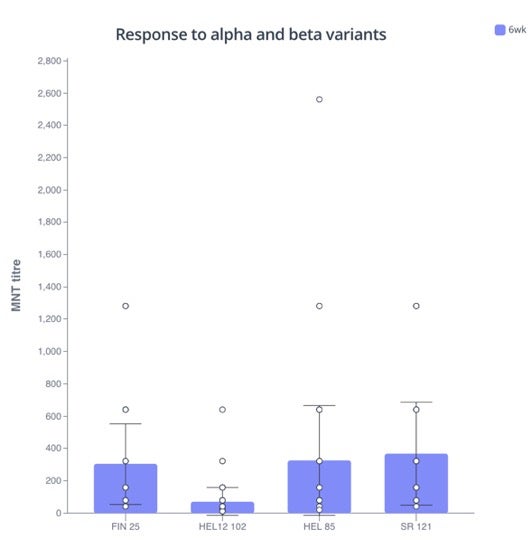
In summary, this study shows that the Pfizer-BioNTech BNT162b2 COVID-19 mRNA vaccine is highly immunogenic and, especially after two vaccine doses, it provides strong protection against the alpha and beta variants.
References
Jalkanen, P., Kolehmainen, P., Häkkinen, H.K. et al. COVID-19 mRNA vaccine induced antibody responses against three SARS-CoV-2 variants. Nat Commun 12, 3991 (2021). https://doi.org/10.1038/s41467-021-24285-4.
Pluto ensures that you not only have fast and reliable results so that you can continue to push the boundaries of knowledge, but that those results are also easily accessible, shareable and publishable right from your browser. Ready to give it a try? Contact us to schedule a personalized demo for your lab.
