Pluto Bioinformatics
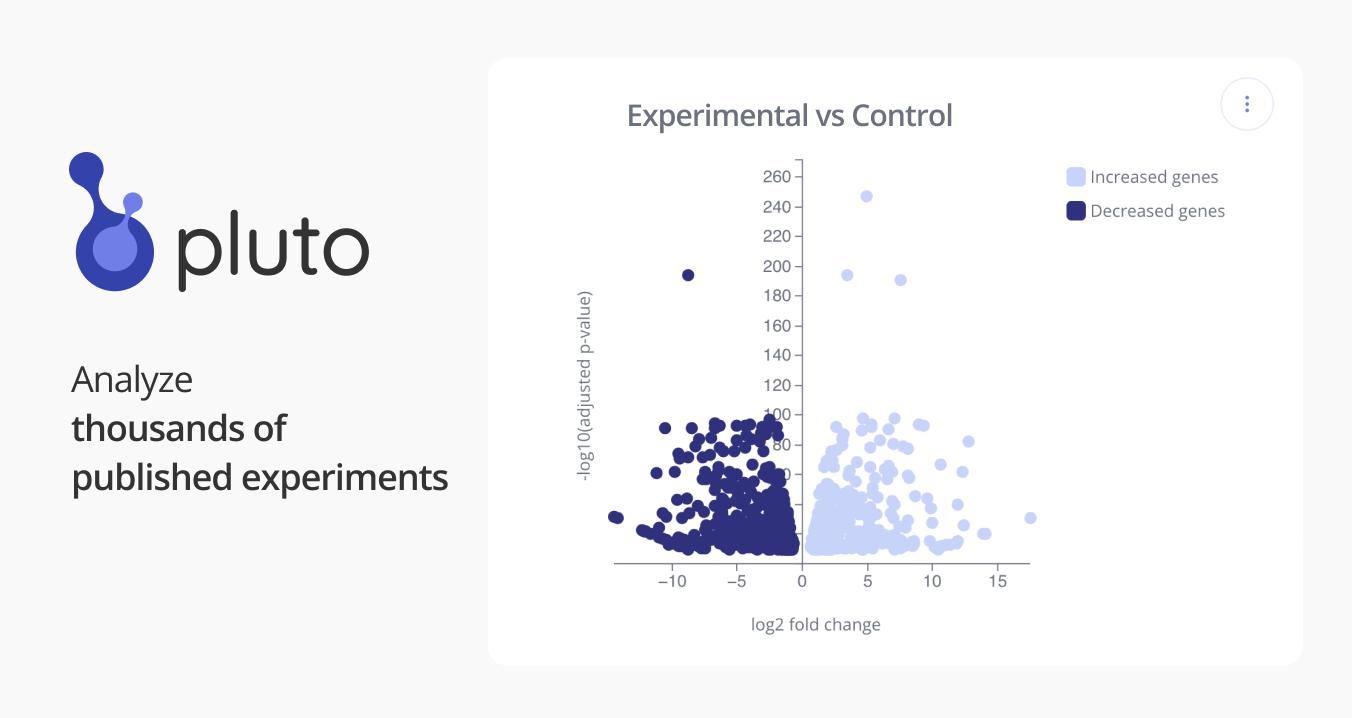
GSE79039: Store-operated Ca2+ entry in follicular T cells controls humoral immune responses and autoimmunity
Bulk RNA sequencing
T follicular helper (TFH) cells promote affinity maturation of B cells in germinal centers (GCs), whereas T follicular regulatory (TFR) cells limit GC reaction. Store-operated Ca2+ entry (SOCE) through Ca2+ release-activated Ca2+ (CRAC) channels mediated by STIM and ORAI proteins is a fundamental signaling pathway in T lymphocytes. Here we show that SOCE is required for the differentiation and function of both TFH and TFR cells. Conditional deletion of Stim1 and Stim2 genes in T cells or Treg cells results in spontaneous autoantibody production and humoral autoimmunity. Conversely, antibody-mediated immune responses following viral infection critically depend on SOCE in TFH cells. Mechanistically, STIM1 and STIM2 control early TFR and TFH cell differentiation through NFAT-mediated IRF4, BATF and Bcl-6 expression. SOCE plays a dual role in GC response by controlling TFH and TFR cell function, thus enabling protective B cell responses and preventing humoral autoimmunity. SOURCE: Martin Vaeth (martin.vaeth@nyumc.org) - Feske Lab New York University